3. 结构
3.1 二维结构
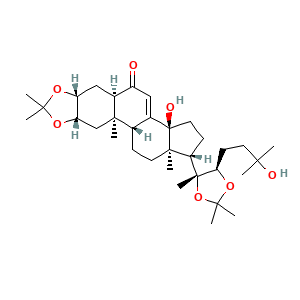
3.2 三维结构
-1
-2
-3
92 97 0 1 0 0 0 0 0999 V2000
-0.1447 0.6850 -1.6726 O 0 0 0 0 0 0 0 0 0 0 0 0
3.4334 -1.9628 0.8588 O 0 0 0 0 0 0 0 0 0 0 0 0
5.5903 -1.0568 0.6782 O 0 0 0 0 0 0 0 0 0 0 0 0
-5.9416 2.3268 1.1786 O 0 0 0 0 0 0 0 0 0 0 0 0
-6.8289 0.9351 -0.4875 O 0 0 0 0 0 0 0 0 0 0 0 0
-4.1954 -3.0369 -1.9924 O 0 0 0 0 0 0 0 0 0 0 0 0
6.0683 3.6934 -1.2903 O 0 0 0 0 0 0 0 0 0 0 0 0
0.7429 -0.6144 0.2264 C 0 0 1 0 0 0 0 0 0 0 0 0
-0.0830 -0.6360 -1.0885 C 0 0 1 0 0 0 0 0 0 0 0 0
2.1579 -0.2935 -0.3192 C 0 0 2 0 0 0 0 0 0 0 0 0
-2.2162 -0.1100 0.2377 C 0 0 2 0 0 0 0 0 0 0 0 0
-3.6381 -0.6502 0.6507 C 0 0 2 0 0 0 0 0 0 0 0 0
0.1646 0.4704 1.1499 C 0 0 0 0 0 0 0 0 0 0 0 0
-1.5303 -0.9896 -0.8233 C 0 0 0 0 0 0 0 0 0 0 0 0
0.7981 -1.4920 -2.0134 C 0 0 0 0 0 0 0 0 0 0 0 0
2.2473 -1.1286 -1.6181 C 0 0 0 0 0 0 0 0 0 0 0 0
-1.3119 0.1886 1.4732 C 0 0 0 0 0 0 0 0 0 0 0 0
3.3288 -0.5458 0.6264 C 0 0 2 0 0 0 0 0 0 0 0 0
-4.4295 -1.0810 -0.6121 C 0 0 1 0 0 0 0 0 0 0 0 0
-4.4752 0.4223 1.3901 C 0 0 0 0 0 0 0 0 0 0 0 0
0.7415 -1.9832 0.9633 C 0 0 0 0 0 0 0 0 0 0 0 0
-4.7923 0.0894 -1.5381 C 0 0 0 0 0 0 0 0 0 0 0 0
4.6728 -0.2010 -0.0130 C 0 0 1 0 0 0 0 0 0 0 0 0
-4.8759 1.6194 0.5231 C 0 0 1 0 0 0 0 0 0 0 0 0
-2.1754 -1.9307 -1.5307 C 0 0 0 0 0 0 0 0 0 0 0 0
-5.4705 1.2430 -0.8179 C 0 0 2 0 0 0 0 0 0 0 0 0
-3.5352 -1.8750 1.6019 C 0 0 0 0 0 0 0 0 0 0 0 0
-3.6416 -2.1102 -1.4034 C 0 0 0 0 0 0 0 0 0 0 0 0
3.0691 0.1379 1.9727 C 0 0 0 0 0 0 0 0 0 0 0 0
4.8388 -2.2534 0.9283 C 0 0 0 0 0 0 0 0 0 0 0 0
5.1288 1.2425 0.0888 C 0 0 0 0 0 0 0 0 0 0 0 0
-7.1542 1.8772 0.5474 C 0 0 0 0 0 0 0 0 0 0 0 0
6.5094 1.4418 -0.5522 C 0 0 0 0 0 0 0 0 0 0 0 0
5.1861 -2.7797 2.3147 C 0 0 0 0 0 0 0 0 0 0 0 0
5.1688 -3.2937 -0.1393 C 0 0 0 0 0 0 0 0 0 0 0 0
-8.0563 1.2019 1.5725 C 0 0 0 0 0 0 0 0 0 0 0 0
-7.8580 3.0882 -0.0628 C 0 0 0 0 0 0 0 0 0 0 0 0
7.0088 2.9008 -0.5624 C 0 0 0 0 0 0 0 0 0 0 0 0
8.3539 2.9787 -1.2922 C 0 0 0 0 0 0 0 0 0 0 0 0
7.1450 3.4821 0.8466 C 0 0 0 0 0 0 0 0 0 0 0 0
2.1827 0.7680 -0.6065 H 0 0 0 0 0 0 0 0 0 0 0 0
-2.3556 0.8606 -0.2580 H 0 0 0 0 0 0 0 0 0 0 0 0
0.2470 1.4617 0.6875 H 0 0 0 0 0 0 0 0 0 0 0 0
0.6663 0.5431 2.1131 H 0 0 0 0 0 0 0 0 0 0 0 0
0.6299 -1.2801 -3.0749 H 0 0 0 0 0 0 0 0 0 0 0 0
0.6402 -2.5646 -1.8534 H 0 0 0 0 0 0 0 0 0 0 0 0
2.8247 -2.0509 -1.5189 H 0 0 0 0 0 0 0 0 0 0 0 0
2.7166 -0.5367 -2.4125 H 0 0 0 0 0 0 0 0 0 0 0 0
-1.3389 -0.6417 2.1846 H 0 0 0 0 0 0 0 0 0 0 0 0
-1.7125 1.0562 2.0113 H 0 0 0 0 0 0 0 0 0 0 0 0
-5.3619 -1.5685 -0.2935 H 0 0 0 0 0 0 0 0 0 0 0 0
-3.9530 0.7899 2.2818 H 0 0 0 0 0 0 0 0 0 0 0 0
-5.3797 -0.0528 1.7904 H 0 0 0 0 0 0 0 0 0 0 0 0
1.2279 -2.7768 0.3879 H 0 0 0 0 0 0 0 0 0 0 0 0
1.1950 -1.9442 1.9552 H 0 0 0 0 0 0 0 0 0 0 0 0
-0.2788 -2.3448 1.1302 H 0 0 0 0 0 0 0 0 0 0 0 0
-3.8920 0.4678 -2.0374 H 0 0 0 0 0 0 0 0 0 0 0 0
-5.4498 -0.2824 -2.3345 H 0 0 0 0 0 0 0 0 0 0 0 0
4.7064 -0.5073 -1.0651 H 0 0 0 0 0 0 0 0 0 0 0 0
-4.0241 2.2968 0.4058 H 0 0 0 0 0 0 0 0 0 0 0 0
-1.6828 -2.5369 -2.2817 H 0 0 0 0 0 0 0 0 0 0 0 0
-5.4951 2.1137 -1.4854 H 0 0 0 0 0 0 0 0 0 0 0 0
-4.5106 -2.3616 1.7227 H 0 0 0 0 0 0 0 0 0 0 0 0
-2.8367 -2.6320 1.2329 H 0 0 0 0 0 0 0 0 0 0 0 0
-3.2120 -1.5826 2.6065 H 0 0 0 0 0 0 0 0 0 0 0 0
-0.5759 0.6075 -2.5413 H 0 0 0 0 0 0 0 0 0 0 0 0
3.9955 0.1610 2.5611 H 0 0 0 0 0 0 0 0 0 0 0 0
2.7607 1.1814 1.8560 H 0 0 0 0 0 0 0 0 0 0 0 0
2.3848 -0.4086 2.6220 H 0 0 0 0 0 0 0 0 0 0 0 0
4.3926 1.8968 -0.3903 H 0 0 0 0 0 0 0 0 0 0 0 0
5.2025 1.5350 1.1410 H 0 0 0 0 0 0 0 0 0 0 0 0
7.2477 0.8114 -0.0409 H 0 0 0 0 0 0 0 0 0 0 0 0
6.4494 1.0866 -1.5898 H 0 0 0 0 0 0 0 0 0 0 0 0
4.9507 -2.0339 3.0816 H 0 0 0 0 0 0 0 0 0 0 0 0
4.6101 -3.6798 2.5537 H 0 0 0 0 0 0 0 0 0 0 0 0
6.2549 -3.0015 2.4000 H 0 0 0 0 0 0 0 0 0 0 0 0
6.2313 -3.5582 -0.1163 H 0 0 0 0 0 0 0 0 0 0 0 0
4.5696 -4.2008 -0.0073 H 0 0 0 0 0 0 0 0 0 0 0 0
4.9607 -2.9119 -1.1440 H 0 0 0 0 0 0 0 0 0 0 0 0
-7.5771 0.3130 1.9944 H 0 0 0 0 0 0 0 0 0 0 0 0
-8.2817 1.8739 2.4071 H 0 0 0 0 0 0 0 0 0 0 0 0
-8.9941 0.8686 1.1155 H 0 0 0 0 0 0 0 0 0 0 0 0
-7.2073 3.6019 -0.7790 H 0 0 0 0 0 0 0 0 0 0 0 0
-8.7622 2.7899 -0.6036 H 0 0 0 0 0 0 0 0 0 0 0 0
-8.1238 3.8205 0.7070 H 0 0 0 0 0 0 0 0 0 0 0 0
8.7062 4.0145 -1.3564 H 0 0 0 0 0 0 0 0 0 0 0 0
8.2589 2.6219 -2.3242 H 0 0 0 0 0 0 0 0 0 0 0 0
9.1214 2.3831 -0.7872 H 0 0 0 0 0 0 0 0 0 0 0 0
6.1697 3.5865 1.3334 H 0 0 0 0 0 0 0 0 0 0 0 0
7.5691 4.4925 0.8103 H 0 0 0 0 0 0 0 0 0 0 0 0
7.7908 2.8624 1.4769 H 0 0 0 0 0 0 0 0 0 0 0 0
6.3910 4.6107 -1.2992 H 0 0 0 0 0 0 0 0 0 0 0 0
1 9 1 0 0 0 0
1 66 1 0 0 0 0
2 18 1 0 0 0 0
2 30 1 0 0 0 0
3 23 1 0 0 0 0
3 30 1 0 0 0 0
4 24 1 0 0 0 0
4 32 1 0 0 0 0
5 26 1 0 0 0 0
5 32 1 0 0 0 0
6 28 2 0 0 0 0
7 38 1 0 0 0 0
7 92 1 0 0 0 0
8 9 1 0 0 0 0
8 10 1 0 0 0 0
8 13 1 0 0 0 0
8 21 1 0 0 0 0
9 14 1 0 0 0 0
9 15 1 0 0 0 0
10 16 1 0 0 0 0
10 18 1 0 0 0 0
10 41 1 0 0 0 0
11 12 1 0 0 0 0
11 14 1 0 0 0 0
11 17 1 0 0 0 0
11 42 1 0 0 0 0
12 19 1 0 0 0 0
12 20 1 0 0 0 0
12 27 1 0 0 0 0
13 17 1 0 0 0 0
13 43 1 0 0 0 0
13 44 1 0 0 0 0
14 25 2 0 0 0 0
15 16 1 0 0 0 0
15 45 1 0 0 0 0
15 46 1 0 0 0 0
16 47 1 0 0 0 0
16 48 1 0 0 0 0
17 49 1 0 0 0 0
17 50 1 0 0 0 0
18 23 1 0 0 0 0
18 29 1 0 0 0 0
19 22 1 0 0 0 0
19 28 1 0 0 0 0
19 51 1 0 0 0 0
20 24 1 0 0 0 0
20 52 1 0 0 0 0
20 53 1 0 0 0 0
21 54 1 0 0 0 0
21 55 1 0 0 0 0
21 56 1 0 0 0 0
22 26 1 0 0 0 0
22 57 1 0 0 0 0
22 58 1 0 0 0 0
23 31 1 0 0 0 0
23 59 1 0 0 0 0
24 26 1 0 0 0 0
24 60 1 0 0 0 0
25 28 1 0 0 0 0
25 61 1 0 0 0 0
26 62 1 0 0 0 0
27 63 1 0 0 0 0
27 64 1 0 0 0 0
27 65 1 0 0 0 0
29 67 1 0 0 0 0
29 68 1 0 0 0 0
29 69 1 0 0 0 0
30 34 1 0 0 0 0
30 35 1 0 0 0 0
31 33 1 0 0 0 0
31 70 1 0 0 0 0
31 71 1 0 0 0 0
32 36 1 0 0 0 0
32 37 1 0 0 0 0
33 38 1 0 0 0 0
33 72 1 0 0 0 0
33 73 1 0 0 0 0
34 74 1 0 0 0 0
34 75 1 0 0 0 0
34 76 1 0 0 0 0
35 77 1 0 0 0 0
35 78 1 0 0 0 0
35 79 1 0 0 0 0
36 80 1 0 0 0 0
36 81 1 0 0 0 0
36 82 1 0 0 0 0
37 83 1 0 0 0 0
37 84 1 0 0 0 0
37 85 1 0 0 0 0
38 39 1 0 0 0 0
38 40 1 0 0 0 0
39 86 1 0 0 0 0
39 87 1 0 0 0 0
39 88 1 0 0 0 0
40 89 1 0 0 0 0
40 90 1 0 0 0 0
40 91 1 0 0 0 0
4. 国际命名与标识
4.1 IUPAC Name
(1R,2R,4S,8R,10R,14S,17S,18R)-14-hydroxy-17-[(4R,5R)-5-(3-hydroxy-3-methylbutyl)-2,2,4-trimethyl-1,3-dioxolan-4-yl]-2,6,6,18-tetramethyl-5,7-dioxapentacyclo[11.7.0.02,10.04,8.014,18]icos-12-en-11-one
4.2 InChl
InChI=1S/C33H52O7/c1-27(2,35)13-12-26-32(9,40-29(5,6)39-26)25-11-15-33(36)20-16-22(34)21-17-23-24(38-28(3,4)37-23)18-30(21,7)19(20)10-14-31(25,33)8/h16,19,21,23-26,35-36H,10-15,17-18H2,1-9H3/t19-,21-,23+,24-,25-,26+,30+,31+,32+,33+/m0/s1
4.3 InChlKey
WXFMGCVRGSIXOB-APTIWFLNSA-N
4.4 Canonical SMILES
CC1(OC2CC3C(=O)C=C4C(C3(CC2O1)C)CCC5(C4(CCC5C6(C(OC(O6)(C)C)CCC(C)(C)O)C)O)C)C
4.5 lsomeric SMILES
C[C@]12CC[C@H]3C(=CC(=O)[C@H]4[C@@]3(C[C@H]5[C@@H](C4)OC(O5)(C)C)C)[C@@]1(CC[C@@H]2[C@@]6([C@H](OC(O6)(C)C)CCC(C)(C)O)C)O
4.6 SDF文件
5. 波谱数据
5.1 13C核磁共振谱(13C NMR)
5.2 1H核磁共振谱(1H NMR)
5.3 质谱(MS)
5.4 红外光谱(IR)
5.5 紫外/可见光谱(UV/Vis)
6. 相关药材
7. 相关靶点
8. 相关疾病